WBBSE Chapter 8 Organic Chemistry Organic Compounds Are Compounds Of Carbon
In the past when there was little knowledge about chemistry, scientists classified compounds on the basis of their sources.
They thought that substances like starch, sugar, proteins, lipids, acetic acid, etc. could only be obtained from plants and animals (living organisms) and called them organic compounds.
On the other hand, the compounds like salts (NaCI), sulfates (CaSO4), nitrates (KNO3), carbonates (CaCO3), etc.
Are obtained from minerals and non-living organisms and they are called inorganic compounds. Lavoisier also accepted this type of idea.
Later, in 1809, Berzelius explained in his vital force theory that organic compounds are only synthesized by living organisms by some unknown vital force and they cannot be prepared in the laboratory.
This old belief has been changed in 1828 when a German scientist Friedrich Wohler was successful to prepare the first organic compound urea in the laboratory.
WBBSE Notes For Class 10 Physical Science And Environment
He took an inorganic salt ammonium cyanate which after heating converted to urea. For this conversion, no vital force was required.
\(\begin{aligned}& \mathrm{NH}_4 \mathrm{CNO} \text { (aq.) } \stackrel{\Delta}{\longrightarrow}\left(\mathrm{NH}_2\right)_2 \mathrm{CO} \text { (aq.) } \\
& \text { ammonium cyanate } \quad \text { urea (a constituent of urine) } \\
&
\end{aligned}\)
Already Lavoisier proved in 1784 that the main constituent of organic compounds is carbon.
In 1845, Kolbe prepared acetic acid and in 1856, Berthelot prepared methane (bio-gas) in the laboratory.
After that, the old concept of chemistry has been changed. Scientists accepted that all organic compounds essentially contain carbon and “organic chemistry is essentially the chemistry of carbon compounds.”
Exceptions: Oxides of carbon (CO, CO2), metallic carbonates (CaCO3), carbides (AI4CI3), metal cyanides (NaCN), etc. They are inorganic compounds.
Organic compounds: The compounds which contain carbon atoms excluding oxides of carbon, metallic carbonates, bicarbonates, carbides, and cyanides are called organic compounds.
They show some special properties like isomerism and catenation which the inorganic compounds do not show.

Difference between organic and inorganic compounds:
Organic compounds must contain carbon, but inorganic compounds may or may not contain carbon.
Organic compounds do not form ionic bonds.
They contain covalent bonds whereas inorganic compounds (like table salt) mostly contain ionic bonds.
Due to the difference in chemical bonding, certain special/different qualities are seen in or ganic compounds such as-
Most of organic compounds are volatile in nature,
The m.p. and b.p. are relatively lower than those of inorganic compounds,
Organic compounds are usually insoluble in water but soluble in organic solvents like benzene, alcohol, etc.
They are non-electrolytes as they are not ionized in an aqueous or fused state,
They have the property of catenation and isomerism,
Most of them form covalent bonds, due to which they react slowly in comparison to inorganic compounds.
WBBSE Chapter 8 Organic Chemistry Tetravalency And Catenation Property Of Carbon
Tetravalency of carbon: The electronic configuration of 6C is = 2 (K) + 4 (L).
It has 4 valence e¯s in the outermost shell. How does C achieve its stable state (duplet/octet state)? Is it either by losing 4e¯s or gaining 4e¯s?
But it doesn’t do any one of this. Because to lose 4e¯s, it would need extra energy and to gain 4e¯s it would be difficult for 6ps to hold 10e¯s.
So for the C atom, transfer of e¯s is not possible. C atom likes to share its e–s. That’s why, C forms 4 bonds.
C is tetravalent, has valency = 4 i.e. its combining capacity is relatively high. It can combine with 4 other atoms having valency 1 or 2.
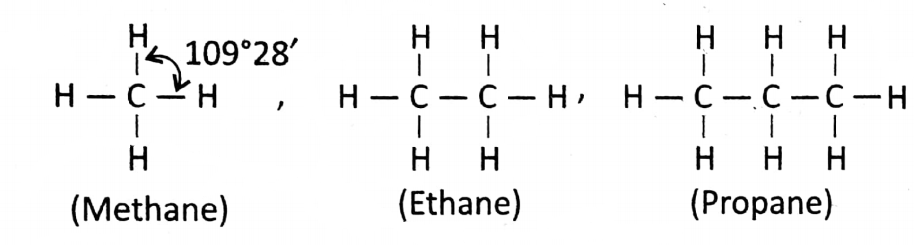
In the simplest organic compound methane molecule, a 1C atom is surrounded by 4 bonds.
Scientist Kekule thought that the structure is planer— something looks like ![]() where the bond angle (angle between 2 bonds) should be 90°.
where the bond angle (angle between 2 bonds) should be 90°.
But this general model couldn’t explain all properties of organic compounds.
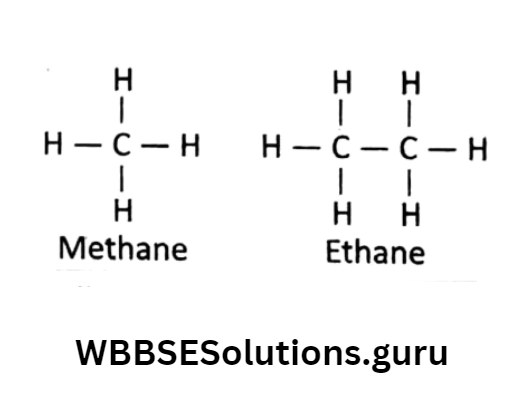
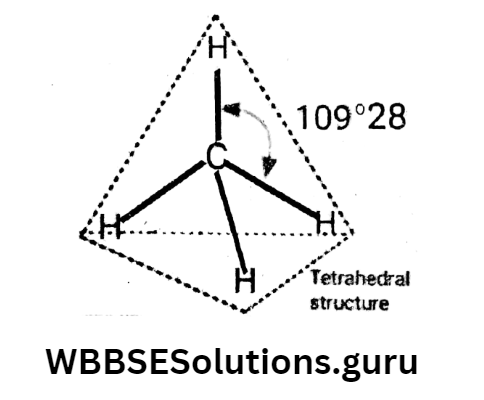
Tetrahedral model: In 1874, Van’t Hoff and Le Bel explained the properties of some organic compounds while trying to rationalize experimental data.
According to this explanation, the structure in methane is tetrahedral (a 3D structure). In case of which all the 4 bonds are not visible.
3 bonds out of 4 are above the plane (towards the observer) and 1 bond is below the plane (away from the observer).
Due to the tetrahedral structure, the bond angle is not 90° it is 109.28′. Later, by X-ray analysis, the model has been proved correct.
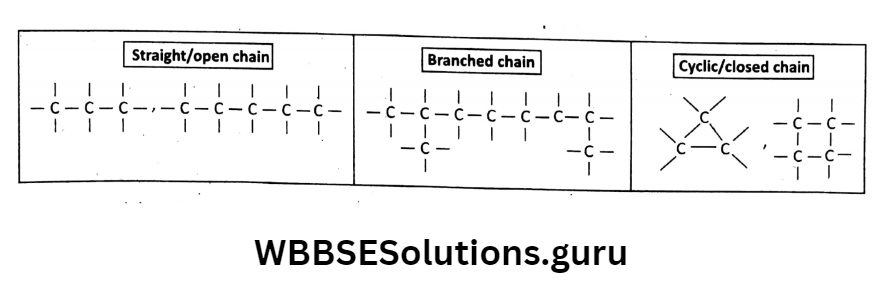
Catenation property of carbon: The self-linking property or tendency of a C-atom to join with another C-atom with a single covalent bond to form a long chain-like structure is called catenation.
Different types of chains can be formed like straight/open chains, branched chains, cyclic closed chains,s or a combination of all these.
For example:
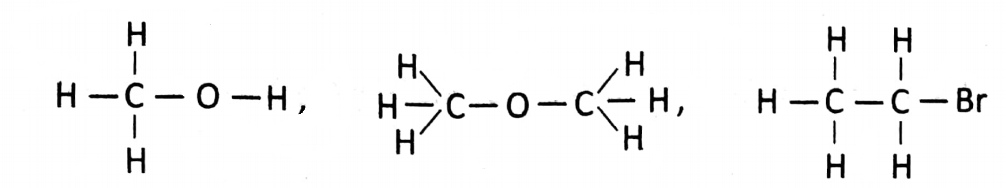
C is tetravalent in nature for which other atoms like another C/H/O/N/F/CI/Br/l/S and so on can join with it and thus different organic compounds are formed.
For example:
Remember:
Most of the time, C and H atoms are joined. C can form bonds in different forms. It could be a single bond/double bond/triple bond.,
(Each covalent bond consists of 2 e¯s).
For example:
(Here the 3D tetrahedral model is projected in the 2D plane).

Structure: Trigonal; Bond angle = 120° (All 3 bonds are visible).
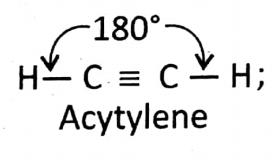
Structure: Coplaner; ingle triple bond; Bond angle = 180° (linear)
WBBSE Chapter 8 Organic Chemistry Structures Of C2H6, C2H4, C2H2
Millions of organic compounds are there. About 10 million C-compounds have already been discovered and still new compounds are being found and synthesized.
Hydrocarbons are the simplest organic compounds.
They contain only C and H atoms—no other elements. The open-chain hydrocarbons are divided into three groups —
- alkanes,
- alkenes and
- alkynes.
(Here ‘o’, ‘e‘, and V are in alphabetical order).
Hydrocarbons are of two types—
Saturated (means less reactive) and unsaturated (very reactive).
The saturated hydrocarbons are those in which the C-atoms are connected by single bonds only (like C – C) while in unsaturated hydrocarbons the C-atoms are connected by at least one double bond (C = C) or triple bond (C = C).
Saturated hydrocarbons are also called alkanes, but unsaturated hydrocarbons are of two types alkenes and alkynes.
Structural and molecular Formula:
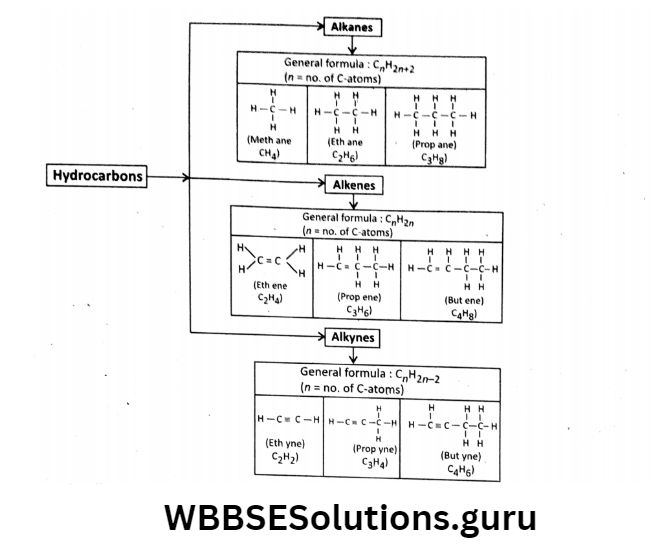
In alkanes, no. of H-atoms = 2 x no.of C-atoms +2; in alkenes, no.of H-atoms = twice the no. of C-atoms, and in alkynes, no. of H- atoms = 2 x no. of C-atoms – 2. For example hex and (n = 6) = I
C6H14 Pent ene (n = 5) = C5H10, hex Yne (n = 6) = C6H10 etc.
Note:
(1) Methane is the simplest alkane, similarly ethene (or ethylene) is the simplest alkene, and ethyne (or acetylene) is the simplest alkyne.
(2) As a whole, the alkanes, alkenes, and alkynes are collectively known as aliphatic hydrocarbons.
WBBSE Chapter 8 Organic Chemistry Functional Groups
We see a wide variety of properties in C-compounds. Because elements other than C and H like 0, N, S, Cl, Br, F … etc. are present in them.
Definition: Functional groups are the groups of atoms that determine all characteristic properties (both physical and chemical) of the compounds in which they are present.
The presence of functional groups determines the properties of C-chains (irrespective of straight or branched).
There are many different functional groups like — OH (alcohol), — CHO (aldehyde), — COOH (carboxylic acid), — NH2 (amine), etc.
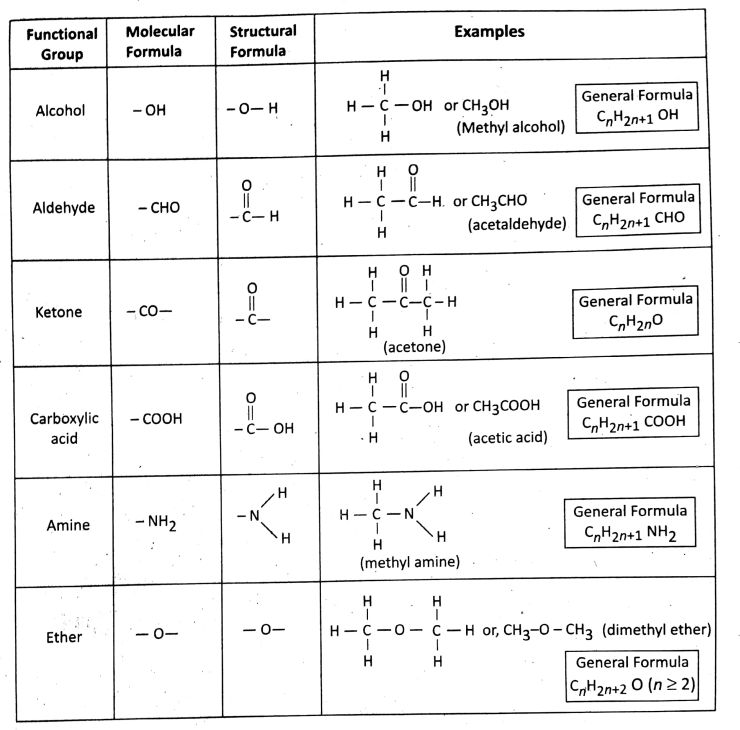
Note: C = C double bond in alkenes and C = C triple bond in alkynes are usually considered functional groups because of which the alkenes and alkynes participate in an addition reaction.
WBBSE Chapter 8 Organic Chemistry Isomerism
The term Isomerism’ originated from two Greek terms Isos→ meaning same or equal and meros → meaning parts. In organic chemistry, isomerism is a very important concept.
There are many C-compounds that have the same molecular formula but different physical and/or chemical properties they are called isomers of each other.
Isomers are made up of the same type of atoms but the atoms are arranged differently.
Basically, two different types of isomerism are seen —
- constitutional isomerism and
- stereoisomerism.
Among these, constitutional isomerism is of different types. Here we will read about two types of constitutional isomerism –
Functional group isomerism and
Positional isomerism.
Functional group isomerism: Same molecular formula but different functional groups having different structures.
Common examples of functional group isomers:
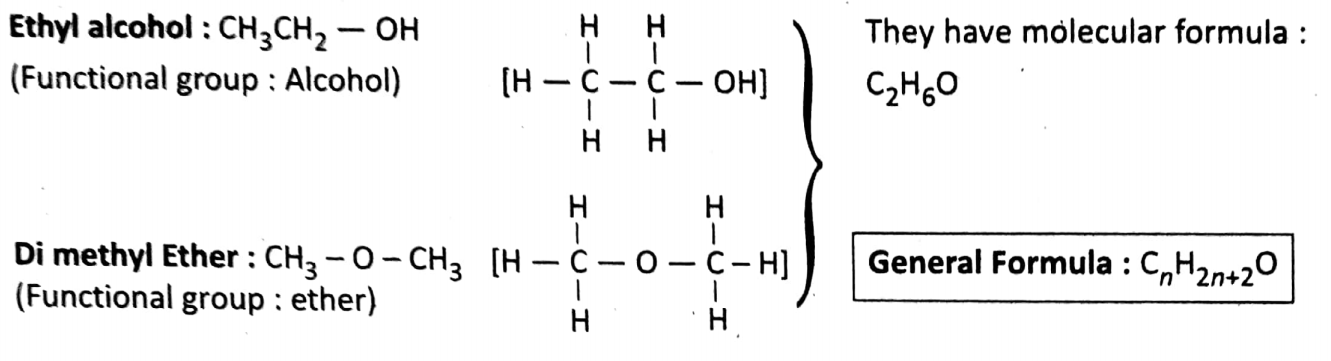
We already got the idea that if functional groups are part of a compound, the entire compound will take the properties of functional groups.
For this reason, functional group isomers show properties different from each other. it is cited here-
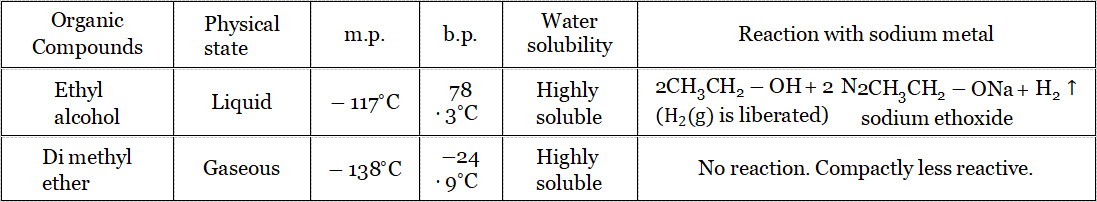
Positional isomerism: In positional isomerism, the difference in structure occurs due to the position of the functional groups must having the same parent chain. Example:

Both have a parent chain of 3 C-atoms.
Molecular Formula: C3HgO In the 1st compound, the functional group (- OH) is attached to the 1st C-atom, whereas in the 2nd compound, with the 2nd C-atom.
So the position of the functional group is different.
Note: Positional isomerism also occurs due to unsaturation (position of double/triple bond in the compound).
WBBSE Chapter 8 Organic Chemistry Homologous Series Homo→Means Same Functional Groups
It is the series of compounds having the same general formula, the same type of molecular structure, and similar chemical properties which appear in immediate succession of molecular mass/Compounds in homologous series (alkanes/alkenes/alkynes) are called homologs.
Characteristic properties of homologous series:
They basically differ in no. of C and H atoms and also molecular mass.
Successive compounds in this series differ by – CH2.
Physical properties like m.p., b.p., and solubility of the compounds that exist in the same series vary in succession.
But the chemical properties are definitely similar as they are dependent on the functional groups present.
The compounds can be artificially prepared by the same method.
For examples:
\(\begin{aligned}& \mathrm{CH}_4 \text { (methane) } \stackrel{+\mathrm{CH}_2}{\longrightarrow} \mathrm{C}_2 \mathrm{H}_6 \text { (ethane) } \stackrel{+\mathrm{CH}_2}{\longrightarrow} \mathrm{C}_3 \mathrm{H}_8 \text { (propane) } \\
& \mathrm{C}_6 \mathrm{H}_{14} \text { (hexane) } \stackrel{+\mathrm{CH}_2}{\longleftarrow} \mathrm{C}_5 \mathrm{H}_{12} \text { (pentane) } \stackrel{+\mathrm{CH}_2}{\longleftarrow} \mathrm{C}_4 \mathrm{H}_{10} \text { (butane) }
\end{aligned}\)

WBBSE Chapter 8 Organic Chemistry IUPAC Nomenclature Of Simple Organic Compounds
Need for IUPAC nomenclature: Carbon has millions and millions of compounds.
Different common names are used for naming organic compounds over the years in different countries.
Common names are like a nickname (what your family members or friends call you). They are not systematic or random.
In 1967, a group of scientists decided to follow a very systematic scheme for naming the organic compounds which are known as IUPAC nomenclature
(Full form: International Union of Pure and Applied Chemistry). IUPAC naming scheme is based on the number of C-atoms in the longest chain/position of functional groups/nature of bonds/…. etc.
Basic IUPAC naming rules:
Root word: First count the number of C-atoms in the longest continuous C-chain. If the compound contains lC-atom, the name starts with meth, for 2C-atoms → eth, 3C -atoms → prop,…. etc. This gives the 1st part of the IUPAC name.
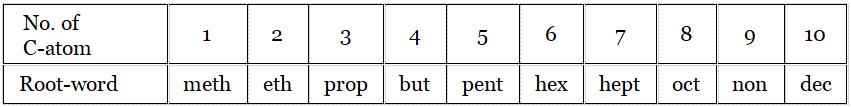
Suffix: Used after root word,
Primary suffix? Check the type of bond between the C-atoms. This gives the 2nd part of the IUPAC name.
Secondary suffix: The name of functional groups comes after the primary suffix.

While combining the IUPAC suffix remove ‘e’ from the alkane,
Make numbering in C-chain forward and backward and chose the lowest possible number for indicating the position of functional groups, Between the number and the letter always use a dash (-). (d) No. 1 in between two parts of the IUPAC name is not necessary.
let us take a few examples:

WBBSE Chapter 8 Organic Chemistry Industrial Source And Major Uses Of CH4, C2H4, C2H2, LPG And CNG
[Methane]→
- Colourless Tasteless Odourless Non-toxic
- Highly flammable
- Abundantly available in marshy areas like wetlands, and swamps where organic substances are decomposed in the absence of O2 by micro-organisms
- Naturally present in natural gas (about 90%)
- second most abundant greenhouse gas after CO2.
Industrial source of CH4:
By destructive distillation of bituminous coal (continuous heating in the absence of air), a gaseous mixture (called coal gas) comes out.
- CH4 occurs in about 30 – 50% of coal gas.
- Biogas or gobar gas contains about 60% CH4.
- CH4 is a major component ( ~90%) of natural gas.
Uses of CH4:
- CH4 is primarily used as a fuel for domestic uses, industries, automobiles, etc.
- Refined liquid CH4 is used as rocket fuel.
- For the production of methyl alcohol, formal dehyde, and methanoic acid by slow combustion (catalytic oxidation) of methane.
- In manufacturing carbon black, printing ink, etc.
[Eythlene]→ Colourless
- Sweat taste and odor
- Natural source of ethylene is petroleum or crude oil.
- Naturally occurring hormone for plants.
Industrial source of C2H4:
By steam cracking of petroleum hydrocarbons at very high temperatures in the presence of a catalyst.
Fractional distillation of petroleum gives a mixture of gases from which C2H4 can be separated.
Uses of C2H4:
- Widely used in the production of polymers like polythene, synthetic rubber, etc.
- Used as a refrigerant, especially in LNG liquefaction.
- Artificial ripening of fruits.
- Oxy-ethylene flame in metal cutting, and welding.
[Eythlene or Acctylene] ⇒
- Colourless, odorless when pure
- Industrial acetylene is
- Extremely explosive
- Produces oxy-acetylene flame (~ 3100°C)
- Non-polluting
Industrial source of C2H2:
- By fractional distillation of crude oil.
- The thermal cracking process by raising the temperature of some suitable hydrocarbons.
- Hydrolysis of calcium carbide.
Uses of C2H2:
- For welding and cutting.
- Used in carbide lamp to get light.
- Production of several inorganic compounds by chemical synthesis of C2H2.
- Used in the production of different variants of plastic like PVC etc.
- (Stands for Liquefied Petroleum Gas) A mixture of (45%) propane (C3H8) and (55%) butane (C4H10)
- Colourless
- Odourless but ethyl mercaptan added to it gives a strong smell for detecting any leakage
- It’s a liquid under high pressure and very low temperature and turns back into gaseous vapor on releasing pressure
- Heavier than air.
Source: LPG is extracted from petroleum/crude oil.
Uses:
- LPG has a higher calorific value (~ 94 MJ/m3) than other conventional fuels and is widely used as cooking gas.
- Also in the petroleum Industry.
CNG⇒ Compressed Natural Gas
Compressed at very high pressure in the cylinders
To avoid transportation problems, it is delivered with the help of pipelines
Composition: Natural gas-basically methane (~ 85 – 90%) with (15 – 10%) ethane, propane, and butane.
Source: Natural gas.
Uses: CNG forms less CO2 and footprints. That’s why it is used as an alternative to petrol/ diesel and can.be used in transportation by bus/taxi.
Advantages of using LPG and CNG: Higher calorific value than conventional fuels like petrol/diesel. Produces no smoke, no soot, and almost no unburnt C-particles.
WBBSE Chapter 8 Organic Chemistry Reactions of CH4, C2H4, C2H2
Some reactions of methane (CH4):
Combustion: Methane is highly flammable but it is not a supporter of combustion.
In a free supply of air (presence of O2), CH4 burns with a non-sooty blue flame and produces CO2 (g) and water vapor.
CH4 + 2O2 CO2 + 2H2O + Heat
The reaction is exothermic (produces a large amount of heat). No C-particles are produced (i.e. pollution-free). That’s why, CH4 can be used as a good fuel.
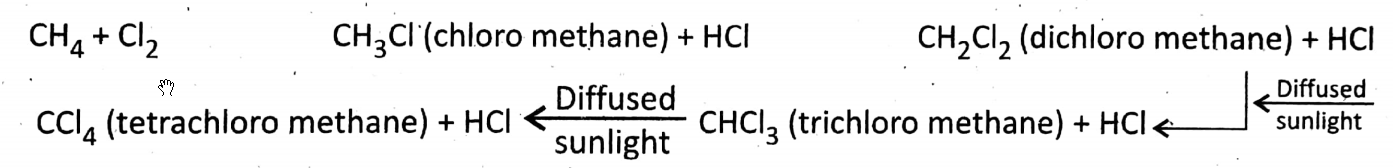
Substitution reaction with chlorine: CH4 is a saturated hydrocarbon for which it is less reactive. It shows only substitution reactions.
In diffused sunlight (not in direct sunlight), CH4 reacts with Cl2 slowly in which 4 H-atoms of CH4 are^ubstituted by Cl-atom and produce different chloro compounds step by step. \(\underset{\text { sunlight }}{\stackrel{\text { Diffused }}{\longrightarrow}}\)
Some reactions of ethylene or ethene (C2H4): It is an unsaturated hydrocarbon (i.e. very much reactive) because of a double bond in it. It takes part in additional reactions.
(1) Addition of H2 or hydrogeneration: At 200°C, in presence of Ni-catalyst, C2H4 reacts with H2 to produce ethane (unsaturated hydrocarbon)

In this reaction, the double (=) bond of C2H4 is broken into a single (-) bond i.e. unsaturated compound is converted into the saturated compound.
Addition reaction with bromine: In the presence of a non-polar solvent like carbon tetrachlo ride (CCI4), Br2 combines with ethylene to produce ethylene di bromide.
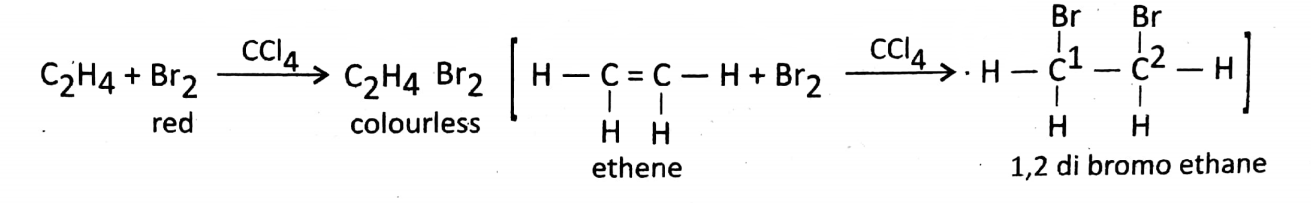
In this reaction, the red color of the bromine solution is decolorized. This reaction can be used as a test for the unsaturated nature of ethene.
Some reactions of acetylene or ethyne (C2H2): It is an unsaturated compound having a triple bond (=). It is also very reactive and does additional reactions.
Addition of H2/hydrogenation: In the presence of Ni-catalyst, at 200°C temperature, C2H2 is added with 2 mol H-, and produces ethane (a saturated compound).

Addition of Br2in the presence of CCI4solvent: This reaction is called
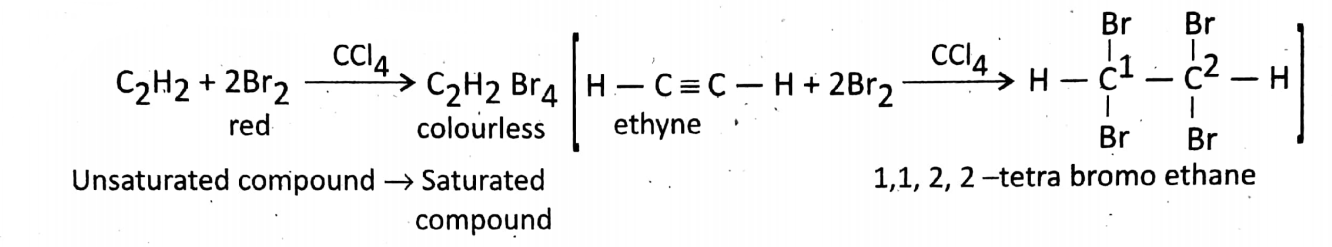
Polymerization of ethylene: Ethene or ethylene is an alkene where 2 C-atoms are joined by a double In this reaction, the red color of the bromine solution is decolorized.
This reaction can be used as a surer test for unsaturated compounds like ethene and ethyne bonds (=).
Polythene or polyethylene is formed when this double bond breaks at a temperature of about 200°C and very high pressure of around 2000 atm in the presence of an organic peroxide (not H2O2) catalyst.
When the double bond breaks, adjacent other molecules link together to form a very large (solid) molecule (macro-molecule) having many small repeating units with very high molar mass.
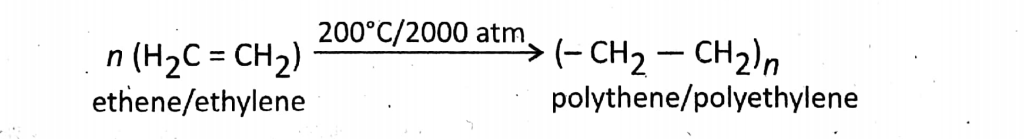
The repeating unit is called the monomer unit and the long molecular chain is called the polymer.
In this reaction, ethylene is the monomer and polyethylene is the polymer.
Homopolymer is made up of only one type of monomer unit, while copolymer is made from two or more types of monomer units.
The process of formation of polymers is called polymerization.
General Difference Between Ethylene and polythylene:

WBBSE Chapter 8 Organic Chemistry Synthetic organic polymers
On the basis of origin, polymers are of two types:
Natural polymers like starch, cellulose, DNA, protein, silk, etc. which are obtained from natural sources,
Synthetic polymers are artificially prepared in the laboratory. Examples of synthetic polymers are plastic, nylon, Teflon, PVC, polystyrene, etc. In our daily life, we use many types of polymers.

WBBSE Chapter 8 Organic Chemistry Biodegradable Polymers
Environmental impact of using synthetic polymers: The biggest problem in using synthetic polymers is that they are not degradable biologically through micro-organisms so the soft drink bottles or the plastic wrappers what we throw away are not biodegradable.
They are not destroyed for hundreds of years. In addition to this, when the rejected plastic materials are burnt toxic fumes directly mix with the atmosphere, the CO2(g) also contributes to global warming.
As a whole, they cause a devastating impact on the environment.
Biodegradable polymers: Materials that are designed such that they get decomposed wholly when exposed to micro-organisms (fungi and bacteria) through aerobic and/or anaerobic processes.
Many natural biodegradable polymers are formed naturally by plants.
Examples: Cellulose, starch, silk, wool, and natural rubber.
Examples of synthetic biodegradable polymers: are bioplastic or green plastic (plant-derived materials), polylactic acid, bio pol resin (PHB + PHV), polyglycolide (polyglycolic and lactic acid,
PHB- Polyhydroxy butyrate; PHV – polyhydroxy venerate).
Long polymer chains are broken down by bacterial action when disposed of without producing toxic substances in the environment.
This is the main advantage of using biodegradable polymers. There are many applications of biodegradable polymers.
Examples: Polyglycolic and lactic acid→heart repair, Dextron→post-operative stitches, Polylactic acid, and lactic acid→drug delivery. –
To avoid non-biodegradable polymers, we can use jute and paper in packing. The use of jute and paper is safe and eco-friendly.
WBBSE Chapter 8 Organic Chemistry Uses And Properties Of Ethyl Alcohol And Acetic Acid
Properties and uses of ethyl alcohol or ethanol (H3C – H2C – OH):
Physical properties:
- Colourless
- Pleasant sweety
- Volatile liquid
- Highly soluble in water
Chemical properties: Ethanol is slightly acidic in nature. It has a tendency to lose H+
Reaction with sodium (a very reactive metal): Ethanol reacts with Na metal vigorously and produces sodium ethoxide (a Na—salt) and releases H2(g).
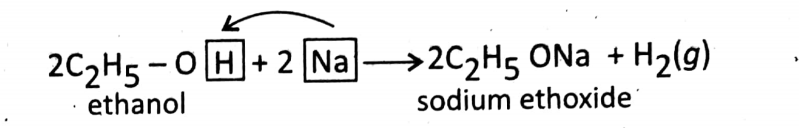
In this reaction, Na replaces H from ethanol (substitution) to produce C2H5ONa.
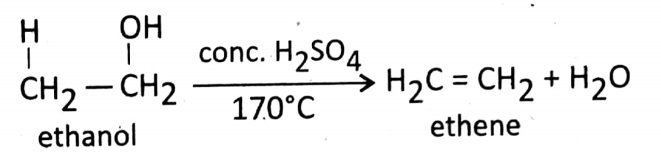
Dehydration reaction (removal of water): At about 170°C temperature, ethanol is dehydrated by the cone. H2SO4(a dehydrating substance).
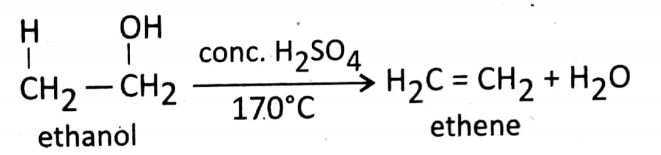
During this reaction, ethanol is converted into ethene.
Uses: Ethanol is used
- As a solvent for gum,
- As antifreeze liquid (due to very low freezing temperature),
- In thermometers,
- In car radiators,
- Mostly for making wine/ beer/whisky.
Properties and uses of acetic acid or ethanoic acid (CH3 – II – OH):
Physical properties: Colourless liquid Characteristic unpleasant pungent odor Soluble in water 5 – 8% solution of acetic acid is called vinegar.
Chemical properties: Less acidic in nature than mineral acids. It has ‘carboxylic acid’ as a functional group.
Reaction with sodium bicarbonate (a weak base):
Reaction with sodium hydroxide (strong base): Ethanoic acid reacts with NaOH to form salt and water (neutralization reaction).

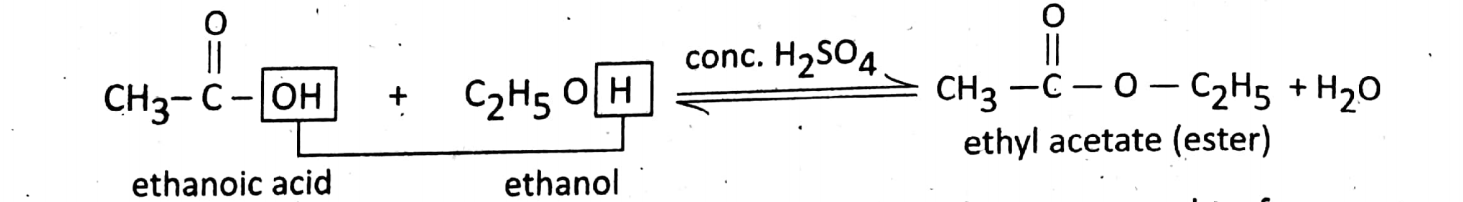
Reaction with ethanol (esterification reaction): The reaction of an alcohol with carboxy lic acid is called esterification which produces a new compound called ester.
In this reaction, OH from ethanoic acid and H from ethanol are removed to form water, and ethyl acetate an ester, having sweet fruity odour is formed.
This reaction establishes the identification of carboxylic group.
Uses: Used
- In manufacturing vinegar
- Esters
- Many polymeric materials like cellulose acetate (used in photographic film)
- As solvent.
Important uses of synthetic polymers:
WBBSE Chapter 8 Organic Chemistry Harmful Effects Of Methanol And Ethanol
Methanol (CH3– OH) is highly poisonous/highjy toxic : It makes alcohol unfit for drinking. If methanol is taken with wine, it oxidises to form toxic formic acid which can cause blind ness, madness or even death.
Ethanol: (CH3 – CH2 – OH) is mainly used in making wine/beer/whisky. In low proportion, it affects cerebral cortex temporarily.
But for excessive intake of ethanol affects the nervous system and harmful for liver/kidney.
WBBSE Chapter 8 Organic Chemistry Denatured Spirit Or Methylated Spirit
In industry, ethanol is used as a solvent. 95% ethyl alcohol is called rectified spirit.
Rectified spirit is denatured by adding toxic substances like methyl alcohol (nearly poisonous), pyride (highly poisonous) and naptha.
This mixture is commercially known as denatured spirit. It is sold excise duty free for industrial use.
In market two types of spirits are available:
Industrial denatured spirit which contains 95% rectified spirit and 5% methyl alcohol,
Mineralised denatured spirit which contains 90% rectified spirit, 9% methyl alcohol and 1% pyride and naptha.;
Uses: Denatured spirit is used as a solvent for varnish.
