WBBSE Chapter 5 Environment Its Resources And Their Conservation Nitrogen Cycle
Introduction to Nitrogen Cycle:
Nitrogen was originally formed in the hearts of stars through the process of nuclear fusion.
When ancient stars exploded, they flung nitrogen-containing gases across the universe. When the earth was formed, nitrogen was a main ingredient in its atmosphere.
The present-day earth’s atmosphere is composed of about 78% nitrogen, about 21% oxygen, and about 1% other gases.
This is an ideal balance because too much Oxygen can be toxic to cells as well as being highly flammable.
Nitrogen, on the other hand, is inert and harmless in its gaseous form.

Nitrogen is one of the primary nutrients critical for the survival of all living organisms since it is a necessary component of many biomolecules such as proteins, DNA, and chlorophyll.
Although nitrogen is very abundant in the atmosphere as dinitrogen gas (N2), it is largely inaccessible in this form to most organisms, making nitrogen a scarce resource and often limiting primary productivity in many ecosystems.
This is because of the strong triple bond between the N atoms in N2 molecules (NN) that makes it relatively inert, whereas organisms need reactive nitrogen to be able to incorporate it into cells.
Read and Learn More WBBSE Solutions For Class 10 Life Science
For plants and animals to be able to use nitrogen, N2 gas must first be converted to a more chemically available form such as ammonium (NH4+), nitrate (NO3), or organic nitrogen (such as urea).
Thus, nitrogen undergoes many different transformations in the ecosystem, changing from one form to another as organisms use it for growth and in some cases, energy.
The movement of nitrogen between the atmosphere, biosphere, and geosphere in different forms is called the nitrogen cycle.
As a major biogeochemical cycle, the nitrogen cycle consists of various reservoirs of nitrogen and processes by which those reservoirs exchange nitrogen.
Nitrogen Cycle Class 10
The nitrogen cycle is the biogeochemical cycle by which nitrogen is converted into multiple chemical forms as it circulates among the atmosphere, and terrestrial and marine ecosystems keeping the balance between the gaseous nitrogen and its form that is useful in biological processes.
The different stages of the nitrogen cycle, which are not altogether sequential, fall into the following classifications:
1. Nitrogen fixation
2. Nitrogen assimilation (ie. nitrogen uptake through organismal growth)
3. Ammonification (ie., nitrogen mineralization through decay)
4. Nitrification
5. Denitrification.
Illustrates the above stages simply:

Microorganisms, particularly bacteria, play major roles in all of the principal nitrogen transformations.
Because these processes are microbially mediated, or controlled by microorganisms, these nitrogen transformations tend to occur faster than geological processes like plate motion which is a part of the carbon cycle.
The rates are affected by environmental factors that influence microbial activity, such as temperature, moisture, and resource availability.
The involvement of prokaryotes in the nitrogen cycle can be shown by a simple.
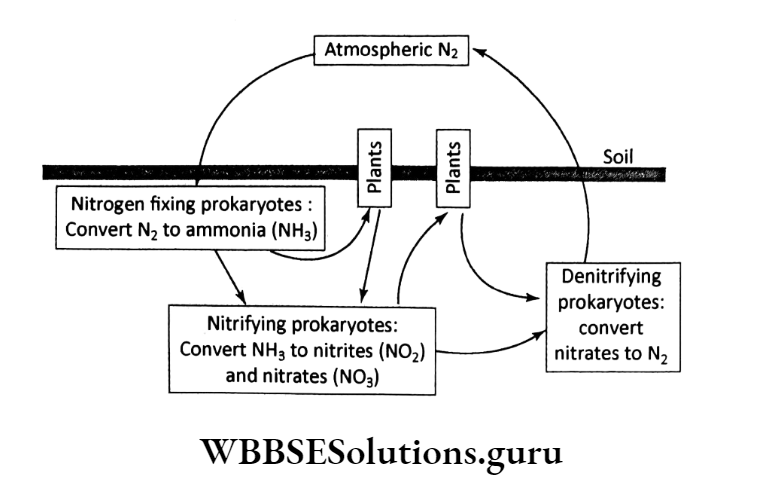
WBBSE Chapter 5 Nitrogen Cycle Nitrogen Fixation
Any natural or industrial process that involves the conversion of atmospheric nitrogen to ammonia and water-soluble nitrites or nitrates which is metabolized by most organisms is referred to as nitrogen fixation.
It can be of three types:
1. Natural or Atmospheric
2. Biological
3. Industrial
About 10% of natural nitrogen fixation takes place by physiochemical methods and 90% by biological methods.
Natural or Atmospheric nitrogen fixation: The fixation of atmospheric nitrogen is a very energy-intensive endeavor.
Under the influence of lightning and thunder, N2 and O2 in the air react to form nitric oxide (NO).
The nitric oxides are again oxidized with oxygen to form different nitrogen oxides.
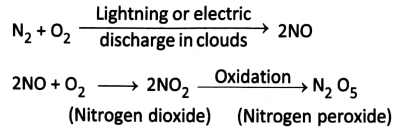
During the rains, NO, combines with rainwater to form nitrous acid (HNO2) and nitric acid (HNO3). The acids fall on the soil along with rain water and react with the alkaline radicals to form water-soluble nitrates \(\left(\mathrm{NO}_3{ }^{-}\right)\) and nitrites \(\left(\mathrm{NO}_2^{-}\right)\)
\(2 \mathrm{NO}_2+\mathrm{H}_2 \mathrm{O} \longrightarrow \mathrm{HNO}_2+\mathrm{HNO}_3(Rainwater)\) \(\mathrm{HNO}_3+\mathrm{Ca} \text { or K Salts } \longrightarrow \text { Ca or K nitrates }
(In soil) \quad (Soluble in water)
\)
The nitrates are soluble in water and as a result, are directly absorbed by the roots of the plants.
Biological nitrogen fixation:
The conversion of molecular nitrogen in the atmosphere into organic nitrogenous compounds through the agency of some living organisms like bacteria and cyanobacteria with the help of an enzyme called nitrogenase is called biological nitrogen fixation.
The action of nitrogenase requires cofactors like iron and molybdenum. Nitrogenase is destroyed in the presence of Oxygen, so these organisms are either anaerobic in nature or they remove oxygen from the site of nitrogenase.
Nitrogen Cycle Class 10
The process of nitrogen fixation involves the conversion of nitrogen to ammonia by gradual reduction. This ammonia is directly used by the organisms to produce complex proteins.
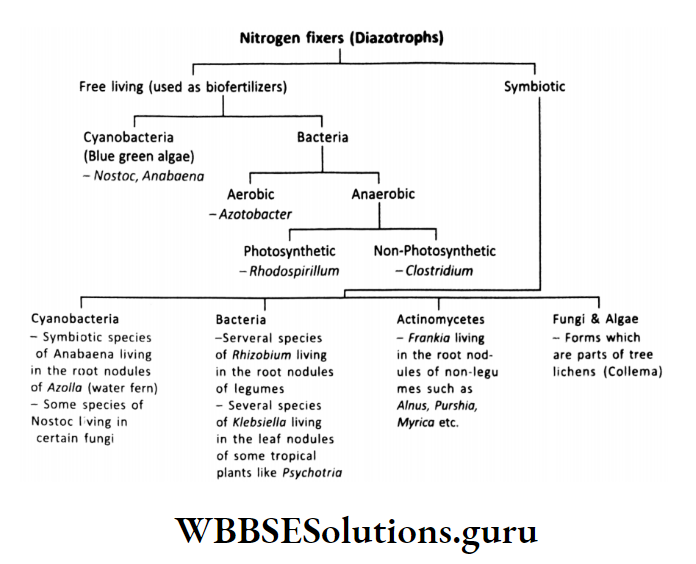
Nitrogen fixers:
Only certain bacteria, some blue-green algae, and leguminous plants Nitrogen fixers:
Only certain bacteria, etc can fix atmospheric nitrogen.
The different types of nitrogen-fixing organisms are represented in the table below:
Mechanism of nonsymbiotic biological N2-fixation:
It requires the presence of hydrogenase and nitrogenase enzyme systems, ferredoxin (a non-heme iron protein) as an electron carrier, pyruvate (an electron donor & energy source), and cofactors like Thiamine Pyro Phosphate, coenzyme-A, inorganic phosphate, and Mg+.
The breakdown of pyruvate produces acetyl phosphate and electrons.
Ferridoxin accepts electrons and gets reduced. [Mechanism of Symbiotic Biological Nitrogen Fixation:]
Acetyl phosphate reacts with ADP to generate ATP.
In the presence of reduced ferredoxin and ATP, nitrogen is absorbed on the surface of the enzyme nitrogenase.
Electrons are now transferred to N2.
Reduction of nitrogen takes place and the enzyme is set free only when nitrogen has been completely reduced to ammonia.
Mechanism Of Symbiotic Biological Nitrogen Fixation:
Symbiotic nitrogen fixation occurs in plants that harbor nitrogen-fixing bacteria within their tissues. The best-studied example is the association between legumes and bacteria in the genus Rhizobium.
Rhizobia are Gram-negative bacilli that live freely in the soil (especially where legumes have been grown). However, they cannot fix atmospheric nitrogen until they have invaded the roots of the appropriate legume.
The Infection Thread
The interaction between a particular strain of rhizobia and the “appropriate” legume is mediated by the “Nod factor” secreted by the rhizobia and transmembrane receptors on the cells of the root hairs of the legume.
Different strains of rhizobia produce different Nod factors, and different legumes produce receptors of different specificity.
If the combination is correct, the bacteria enter an epithelial cell of the root and then migrate into the cortex. Their path runs within an intracellular channel that grows through one cortex cell after another.
This infection thread is constructed by the root cells, not the bacteria, and is formed only in response to the infection.
When the infection thread reaches a cell deep in the cortex, it bursts and the rhizobia are engulfed by endocytosis into membrane-enclosed symbiosomes within the cytoplasm.
At this time the cell goes through several rounds of mitosis without cytokinesis so the cell becomes polyploid. The cortex cells then begin to divide rapidly forming a nodule.
This response is driven by the translocation of cytokinins from epidermal cells to the cells of the cortex.
The rhizobia also go through a period of rapid multiplication within the nodule cells. Then they begin to change shape and lose their motility.
The bacteroids, as they are now called, may almost fill the cell. Only now does nitrogen fixation begin.
Root nodules are not simply structureless masses of cells. Each becomes connected by the xylem and phloem to the vascular system of the plant.
Thus the development of nodules, while dependent on rhizobia, is a well-coordinated developmental process of the plant. In addition, the legume host supplies one critical component of nitrogenase —the key enzyme for fixing nitrogen.
The bacteroids need oxygen to make their ATP (by cellular respiration). However, nitrogenase is strongly inhibited by oxygen. Nodules are filled with hemoglobin.
So much of it, in fact, that a freshly-cut nodule is red.
The hemoglobin of the legume (called leghaemoglobin), like the hemoglobin of vertebrates, probably supplies just the right concentration of oxygen to the bacteroids to satisfy their conflicting requirements.
Wbbse Class 10 Life Science Nitrogen Cycle
The metal molybdenum is a critical component of nitrogenase and so is absolutely essential for nitrogen fixation. The whole process of nitrogen fixation is controlled by nef genes.
Because of the specificity of the interaction between the Nod factor and the receptor on the legume, some strains of rhizobia will infect only peas, some only clover, some only alfalfa, etc.
The treating of legume seeds with the proper strain of rhizobia is a routine agricultural practice.
Industrial Nitrogen Fixation:
The industrial production of ammonia is carried out by mixing nitrogen and hydrogen at high temperatures and pressure (i.e. about 150-250 millibar and 400- 500°C).
The process known as the Haber-Bosch process, requires an iron catalyst (magnetite or Fe304) which removes oxygen from the environment.
\(\mathrm{N}_2+3 \mathrm{H}_2 \rightleftharpoons 2 \mathrm{NH}_3\)WBBSE Chapter 5 Nitrogen Cycle Nitrogen Assimilation
Nitrogen assimilation is the formation of organic nitrogen compounds like amino acids from inorganic nitrogen compounds present in the environment.
Plants absorb nitrogen from the soil in the form of nitrate (NO3+) and ammonium (NH4+). In aerobic soils, nitrate is usually the predominant form of available nitrogen that is absorbed.
Ammonia can predominate in grasslands and in flooded, anaerobic soils. Both ammonium and nitrate are taken up by plants by using several transporters.
Nitrogen is transported from the root to the shoot via the xylem in the form of nitrate, dissolved ammonia, and amino acids.
Nitrate is reduced to nitrite by the enzyme nitrate reductase and nitrite is then reduced to ammonia in a series of two-electron transfers by nitrite reductase.
Ammonia (both absorbed and synthesized) is incorporated into amino acids via the GS- GOGAT pathway.
\(\mathrm{NO}_3^{-} \underset{2 e^{-}}{\longrightarrow} \mathrm{NO}_2^{-} \underset{6 e^{-}}{\longrightarrow} \mathrm{NH}_3\)WBBSE Chapter 5 Nitrogen Cycle Ammonification
The process is also called nitrogen mineralization, which involves the conversion of complex organic nitrogenous compounds present in the dead bodies of plants and animals to ammonium ions.
It is essentially carried out by an array of bacteria present in the soil like Bacillus ramosus, Bacillus vulgaris, Bacillus my codes, etc. in the presence of enzymes like GS, GOGAT, GDH, etc.
The ammonia released in the soil changes the soil pH and reduces the toxic acidophilic bacteria. This ammonia becomes the substrate for the nitrifying bacteria, which ultimately increases soil fertility.
Amino acid + Oxygen→ Keto acid + Ammonia
Wbbse Class 10 Life Science Nitrogen Cycle
WBBSE Chapter 5 Nitrogen Cycle Nitrification
The stagewise microbial conversion of ammonium ion to nitrite and then nitrite to nitrate is called nitrification.
The process occurs in two steps.
The first step is Nitritation, carried out by Nitrosomonas. \(\mathrm{NH}_3+\mathrm{O}_2 \rightarrow \mathrm{NO}_2+\mathrm{H}^{+}+\mathrm{H}_2 \mathrm{O}\)
The second step is called Nitratation, carried out by Nitrobacter. \(\mathrm{NO}_2+\mathrm{O}_2 \rightarrow \mathrm{NO}_3\)
Some soil conditions controlling nitrification rates are:
- Substrate availability (ie., presence of NH4+)
- Availability of 02 (c) Soil drainage
- Soil pH
- Soil temperature
WBBSE Chapter 5 Nitrogen Cycle Denitrification
The process involves the reduction of soil nitrate to nitrogen gas by bacteria like Bacillus denitrificans, Thiobacillus denitrificans, Paracoccus denitrificans, Micrococcus, Thiobacillus thiolates, Pseudomonas, etc.
Denitrifying microbes require a very low oxygen concentration as well as organic C for energy. More than one enzymatic pathway has been identified in the reduction process.
The denitrification process may generate a series of oxides of nitrogen, which also includes N2O, which is a greenhouse gas and may cause global warming.
\(2 \mathrm{NO}_3^{-} \rightarrow 2 \mathrm{NO}_2 \rightarrow 2 \mathrm{NO} \rightarrow \mathrm{N}_2 \mathrm{O} \rightarrow \mathrm{N}_2\)Steps Of Nitrogen Cycle Class 10
Direct reduction from nitrate to ammonium compounds is possible, which is also called dissimilatory nitrate reduction (DNRA), which is carried out by bacteria like Alcaligenes, Bradyrhizobium, etc.
Some genes known in microorganisms that denitrify include nir, nos, etc.
WBBSE Chapter 5 Nitrogen Cycle Diagrammatic Sketch Of The Nitrogen Cycle

WBBSE Chapter 5 Nitrogen Cycle Significance Of The Nitrogen Cycle
There are several importance of the nitrogen cycle which are mentioned below:
The nitrogen cycle brings in the inert nitrogen from the air into the biochemical process in plants and then to animals.
Plants need nitrogen to synthesize chlorophyll and so the nitrogen cycle is absolutely essential for them.
During ammonification, the bacteria help degrade decomposing animal and plant matter. This helps in naturally cleaning up the environment.
Due to the nitrogen cycle, nitrates and nitrites are released into the soil which helps in enriching the soil with nutrients needed for cultivation.
Animals obtain nitrogenous compounds from plants. Nitrogen is needed as an integral part of cell composition. It is due to the nitrogen cycle that animals are also able to utilize the nitrogen present in the air.
WBBSE Chapter 5 Nitrogen Cycle Human Activities And Nitrogen Cycle
Human activities have greatly influenced the nitrogen cycle. The increase in the global population has necessitated an increase in the production of food.
This is mainly done by the addition of nitrogen-enriched fertilizer including urea in the soil, which has resulted in the liberation of more nitrogenous compounds in nature. This has affected the global ecosystem in the following ways:
The increased generation of nitrous oxide (N2O) results in global warming and increased melting of permafrost causing elevation of water levels in the sea and ocean.
Elevation in the level of NO and NO2 due to the burning of fossil fuel results in acid rain, which damages the leaf surface reducing the photosynthetic productivity, reducing soil pH and fertility of the soil, and damaging buildings and monuments.
This increases the population of acidophilic bacteria, which may cause pathogenicity in animals including human beings.
Steps Of Nitrogen Cycle Class 10
The reduction in the pH of water bodies including streams and lakes and the discharge of agricultural runoff containing excess nitrates cause the destruction of aquatic fauna including fish.
The acidic pH of water damages the pipeline and also the drainage system.
WBBSE Chapter 5 Nitrogen Cycle Fill In The Blanks
Question 1. The percentage of Nitrogen in the atmosphere is____________________
Answer: 78%
Question 2. The bio-geochemical cycle include oxygen, carbon and____________________ .
Answer: Nitrogen
Question 3. The protein sub units are called____________________ .
Answer: Amino acid
Question 4. Nostoc is a____________________ that fixes nitrogen.
Answer: Cyanobacteria
Question 5. The percentage of nitrogen in protein is____________________ .
Answer: 16%
Question 6.____________________ is synthesized during lightning.
Answer: No
Nitrogen Cycle Class 10 MCQs
Question 7. Ammonia to nitrate conversion is called____________________.
Answer: Nitrification
Question 8. NO to NO2 conversion occurs by____________________ .
Answer: Oxidation
Question 9. Leg-haemoglobin transports____________________ .
Answer: Oxygen
Question 10. ____________________ causes ammonification.
Answer: Bacillus mycoides
Question 11. HN03 reacts with calcium of rock to produce____________________ .
Answer: calcium nitrate
Question 12. During acid rain, the common acids formed are sulfurous and ____________________acid.
Answer: Nitrous
Question 13. The Alnus nodule contains____________________ .
Answer: Frankia
Question 14. Nitrogen is____________________ to ammonia during .
Answer: Reduced Nitrogen fixation
Question 15. ____________________ is a denitrifying bacteria.
Answer: Pseudomonas
Question 16. The smog is created by which forms ____________________.
Answer: NO2 Pan (Peroxy Acettl Nitrate)
WBBSE Chapter 5 Nitrogen Cycle Match The Column

Answer: 1-C,2-D,3-A,4-B

Answer: 1-C,2-D,3-A,4-B
WBBSE Chapter 5 Nitrogen Cycle Write True Or False
Question 1. Nitrogen is directly absorbed from nature by higher organisms.
Answer: False
Question 2. The plant absorbs nitrate from the soil.
Answer: True
Question 3. Pseudomonas helps in nitrification.
Answer: False
Question 4. Fertilizer contains N, P, and K.
Answer: True
Question 5. Protoplasm contains inorganic nitrogen.
Answer: False
Question 6. Bacillus ramosus is a nitrifying bacteria.
Answer: False
Question 7. Anabaena is a nitrogen-fixing bacteria.
Answer: False
Question 8. Thiobacillus is a denitrifying bacteria.
Answer: True
Nitrogen Cycle Class 10 MCQs
Question 9. The atmosphere contains 78% nitrogen
Answer: True
Question 10. Nitrogen dioxide is a greenhouse gas.
Answer: False
Question 11. Azotobacter is a free-living nitrogen-fixing bacteria.
Answer: True
Question 12. Oxygen is a poison for nitrogenase enzymes.
Answer: True
WBBSE Chapter 5 Nitrogen Cycle Very Short Answer Type Questions
Question 1. Name endophytic cyanobacteria.
Answer: Anabaena azollae.
Question 2. Name two free-living nitrogen-fixing bacteria.
Answer: Azotobacter and Clostridium.
Question 3. Which bacteria convert Ammonia to nitrite?
Answer: Nitros monas.
Question 4. Name the most important compound containing nitrogen in the cytosol.
Answer: Amino acid.
Question 5. Name the enzyme that fixes atmospheric nitrogen.
Answer: Nitrogenase.
Question 6. Name the red-colored pigment present in the nodule of the leguminous plant.
Answer: Leg-haemoglobin (Lb).
Question 7. What amount of ATP is needed to fix one molecule of N2?
Answer: 16 ATP.
Question 8. What is the name of a nitrogen-fixing gene?
Answer: if gene.
Question 9. Name two gram-ve nitrogen-fixing bacteria.
Answer: Klebsiella and Rhizobium.
Question 10. Name the co-factor for nitrogenase.
Answer: Fe and Mo.
Nitrogen Cycle Class 10 MCQs
Question 11. What are diazotrophs?
Answer: The nitrogen-fixing bacteria and archaea are together called diazotrophs.
Question 12. State the equation for biological nitrogen fixation.
Answer: \(\mathrm{N}_2+8 \mathrm{H}^{+}+8 \mathrm{e} \rightarrow 2 \mathrm{NH}_3+\mathrm{H}_2\)
Question 13. Name a marine cyanobacterium fixing nitrogen.
Answer: Trichodesmium.
Question 14. Name 2 nitrogen-fixing lichen.
Answer: isobaric and Peltigera.
Question 15. Name 2 plants showing actinorhiza.
Answer: Ain us sp and Myrica sp.
Question 16. A pair of related terms is given below. Based on the relationship in the first pair, write the suitable word in the gap of the second pair.
Answer: Rhizobium: root nodule:: Klebsiella: leaf nodules
Steps Of Nitrogen Cycle Class 10
Question 17. Which one is the most predominant method of nitrogen fixation?
Answer: Biological nitrogen fixation
Question 18. Give examples (one each) of legumes that are poor fixers and good fixers of nitrogen.
Answer: Poor fixer—Common beans Good fixer—Soybeans
WBBSE Chapter 5 Nitrogen Cycle Short Answer Type Questions
Question 1. What is the nitrogen cycle?
Answer:
Nitrogen cycle
The nitrogen cycle is the biogeochemical cycle by which nitrogen is converted into multiple chemical forms as it circulates among the atmosphere, and terrestrial and marine ecosystems keeping the balance between the gaseous nitrogen and its form that is useful in biological processes.
Question 2. What is ammonification?
Answer:
Ammonification
Ammonification, also called nitrogen mineralization, involves the conversion of complex organic nitrogenous compounds present in the dead bodies of plants and animals to ammonium ions by an array of bacteria present in soil like Bacillus ramosus, Bacillus vulgaris, etc.
Amino acid + Oxygen→ Ketoacid + Ammonia
Nitrogen Cycle Diagram Class 10
Question 3. What is nitrification?
Answer:
Nitrification
The biological degradation of ammonia to nitrite and nitrite to nitrate by bacteria like Nitrosomonas and Nitrobacter is called nitrification.
\(\begin{aligned}& \mathrm{NH}_3+\mathrm{O}_2 \rightarrow \mathrm{NO}_2+\mathrm{H}^{+}+\mathrm{H}_2 \mathrm{O} \\
& 2 \mathrm{NO}_2+\mathrm{O}_2 \rightarrow 2 \mathrm{NO}_3
\end{aligned}\)
Question 4. What is denitrification?
Answer:
Denitrification
The biological conversion of soil-borne nitrite or nitrate to nitrogen gas by bacteria like Pseudomonas is called denitrification.
\(2 \mathrm{NO}_3^{-} \rightarrow 2 \mathrm{NO}_2 \rightarrow 2 \mathrm{NO} \rightarrow \mathrm{N}_2 \mathrm{O} \rightarrow \mathrm{N}_2\)Question 5. What is Haber-Bosch process?
Answer:
Haber-Bosch process
The chemical process of generating ammonia by mixing nitrogen and hydrogen under high temperatures (400-500 °C) and pressure (about 150-250 millibar) in the presence of an iron catalyst is called the Haber-Bosch process.
\(\mathrm{N}_2+3 \mathrm{H}_2 \rightleftharpoons 2 \mathrm{NH}_3\)Question 6. What are the constituents of NOx?
Answer:
The constituents of NOx
NO, NO2, and N2O.
Question 7. What is the role of diazotrophs in the nitrogen cycle?
Answer:
The role of diazotrophs in the nitrogen cycle
The diazotrophs are bacteria containing molybdenum or vanadium nitrogenase enzyme systems which combine nitrogen and hydrogen to produce biologically useable ammonia under normal temperature and pressure. Thus diazotrophs facilitate the consumption of nitrogen by plants by fixing them.
Question 8. How does Rhizobium develop symbiotic associations with legume plants?
Answer:
Inside the root nodules of leguminous plants, Rhizobium differentiates into bacteroids and fixes atmospheric nitrogen into ammonium using the enzyme nitrogenase.
Ammonium is then converted to amino acids before it is exported to the plant. In return, the plant supplies the bacteria with carbohydrates in the form of organic acids.
The plant also provides the bacteroid oxygen for cellular respiration, tightly bound by leghaemoglobins.
This way the symbiotic relationship of Rhizobium develops with the legume plants involving a signal exchange between both partners and the development of symbiotic structures.
Question 9. What is the role of nitrogen in the legume plant?
Answer:
The role of nitrogen in the legume plant
Nitrogen is used for protein and enzyme synthesis, amino acid and nucleic acid, and chlorophyll biosyntheses.
Thus plants with sufficient nitrogen experience high rates of photosynthesis and typically exhibit vigorous plant growth and development.
Question 10. What is biofertilizer?
Answer:
Biofertilizer
A substance containing the nitrogen-fixing free-living bacteria like Azatobacter, and Clostridium and cyanobacteria like Nostoc, and Anabaena that increases soil fertility through nitrogen fixation and thereby promotes growth by increasing the supply or availability of primary nutrients to the plant is called biofertilizer.
Biofertilizers provide eco-friendly organic agro-input since the microorganisms in them restore the soil’s natural nutrient cycle and build soil organic matter.
Importance Of Nitrogen Cycle Class 10
Question 11. Name the acids formed from nitrogenous oxides.
Answer:
Nitrous acid and nitric acid are produced from oxides of nitrogen like nitric oxide and nitrogen dioxide.
Question 12. What is Leghaemoglobin?
Answer:
Leghaemoglobin
Leghaemoglobin is an oxygen carrier and hemoprotein found in the nitrogen-fixing root nodules of leguminous plants.
It is produced by legumes only in response to the roots being colonized by nitrogen-fixing rhizobia as part of the symbiotic interaction between the plant and the bacteria.
The holoprotein (protein + heme cofactor) is red in color, has close chemical & structural similarities to hemoglobin, and has a high affinity for oxygen.
Question 13. List two main human activities that alter the nitrogen cycle.
Answer:
Two main human activities that alter the nitrogen cycle in both aquatic and terrestrial ecosystems are-
- Burning of fossil fuels
- Application of nitrogen-based fertilizers in agriculture which often ends up in a runoff
Question 14. List out the sources of nitrogen to plants. Sources of nitrogen to plants:
Answer:
- Atmospheric molecular nitrogen
- Nitrates, nitrites & ammonia in the soil
- Aminoacids (organic nitrogen) in soil
- Small insects (in the case of insectivorous plants)
