WBBSE Chapter 8 Metallurgy Uses Of Fe, Cu, Zn, And Al And Their Alloys

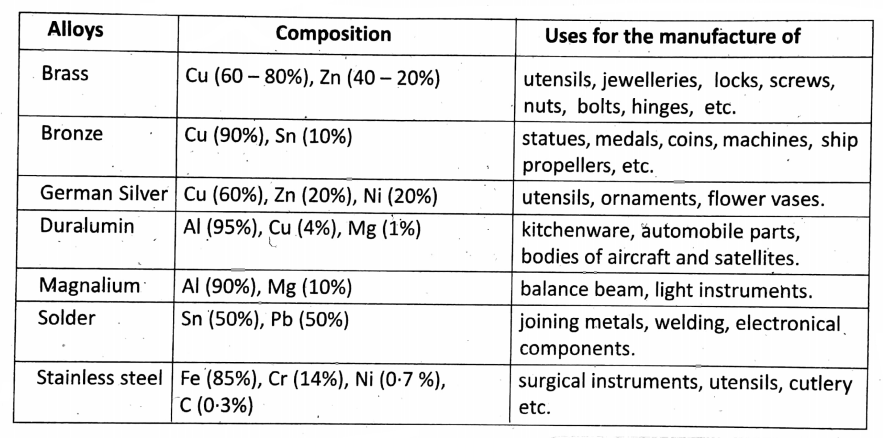
Advantages of using alloys:
Alloys are harder than their constituent metals but less ductile and malleable. Example: Aluminium is lightweight, but its alloy duralumin (Al + Cu + Mg + Mn) is light but strong. Copper is soft, but it is alloy brass and bell metal are strong.
WBBSE Notes For Class 10 Physical Science And Environment
Alloys are resistant to corrosion: For example, Iron rusts, but stainless steel (Fe + C) does not. The melting point of an alloy may be lower than any of its original constituents.
Example: For soldering, an alloy of Pb and Sn is used whose m.p. is lower than that of Pb or Sn. Using alloys the electrical conductivity can be changed.
Example: The alloy of Ni + Cr + Fe, called nichrome, resistivity is high.
In making 24-carat pure gold jewellery, 22 parts of gold are mixed with 2 parts of either Cu or Ag. So the jewellery we use is alloys.
Some important alloys:
WBBSE Chapter 8 Metallurgy Ores And Minerals
Most of the metals exist in the earth’s crust in combined states with some impurities such as sand/stones/rocks/limestone.
These naturally occurring substances in the earth’s crust which contain metals are called minerals. For example, iron exists in the earth’s crust as sulphides/carbonates/oxides/nitrates, with some impurities like C, Si, S, Mn etc.
Metallurgy refers to the process of extraction of pure metals from some minerals (ores). But from all minerals, metals cannot be extracted.
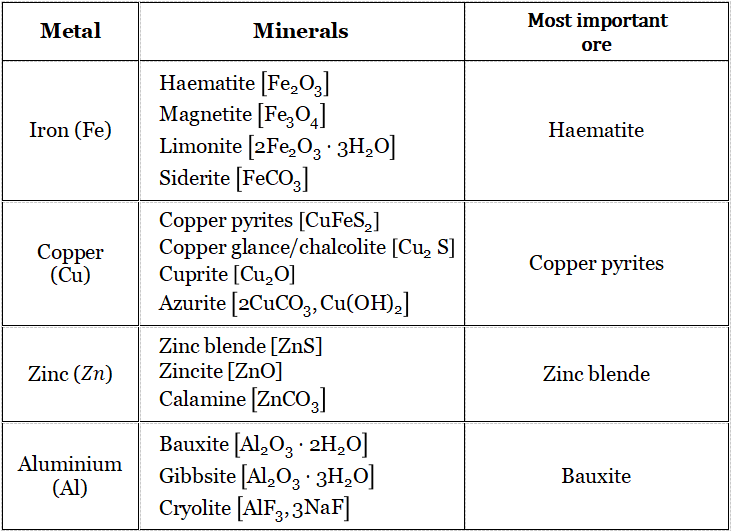
Minerals from which pure metals can be extracted cheaply and easily/conveniently are called ores. For example,
Al is most abundant in the earth’s surface /clay. But Al is not extracted from clay. Because the process is very costly. Rather, Al is extracted from bauxite.
So that bauxite is the ore of Al.
Haematite (Fe2O3) and iron pyrites (FeS2) both contain a high percentage of Fe. However the removal of S from FeS2 is very difficult and costly also. So FeS2 is not considered as the ore of Fe.
Conclusion: All minerals are not ores, but all ores are minerals.
Major ores of Fe, Cu, Zn and Al :

WBBSE Chapter 8 Metallurgy Electronic Theory And Redox Processes
According to electronic theory, oxidation is loss of e~ and reduction is gain in e¯.
Examples of oxidation: \(\mathrm{Na}-e^{-} \rightarrow \mathrm{Na}^{+} ; \mathrm{Ca}-2 e^{-} \rightarrow \mathrm{Ca}^{2+}\)
Examples of reduction: \(\mathrm{Al}^{3+}+3 \mathrm{e}^{-} \rightarrow \mathrm{Al} ; \quad \mathrm{Fe}^{3+}+3 e^{-} \rightarrow \mathrm{Fe} ; \quad \mathrm{Zn}^{2+}+2 e^{-} \rightarrow \mathrm{Zn}\)
In metallurgy reduction/dressing/removal of oxygen of metal oxide is a very important step.
In order to free metal from its oxide, the reduction is done by excluding the non-metallic part Usually, the reduction is done in two ways:
Oxides of Cu, Pb, and Fe can be reduced by using chemical-reducing agents like C (coke), CO, H2, and NH3.
ZnO can only be reduced using coke (C) heated at high temperatures.
⇒ \(\mathrm{ZnO}+\mathrm{C} \rightarrow \mathrm{Zn}+\mathrm{CO}\) (carbon reduction of ZnO) Reduction
Note: During electrolytic extraction, reduction takes place at the cathode. General equation:
Mn++ ne¯→ M (where M = metal, n = number of electrons)

WBBSE Chapter 8 Metallurgy Thermite Reaction
Al has a high affinity to react with O2 at high temperatures and the reaction is exothermic in nature. Using this principle, Fe is extracted from its oxide using the Goldschmidt thermite process.
Thermite mixture is basically a mixture of fine aluminium oxide (Al2O3) and ferric oxide (Fe2 O3) in a ratio of 1: 3 by mass.
The mixture also consists of a very minimal amount of igniting material like barium peroxide (BaO2).
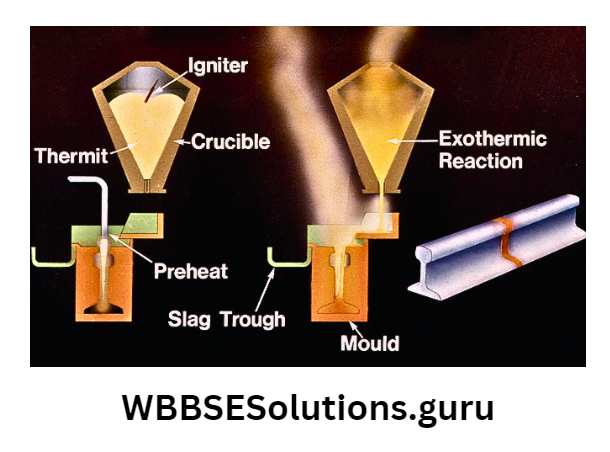
Taking the whole mixture in a crucible, ignite it with an Mg-ribbon. As soon as Mg- ribbon is burnt, a high temperature is created. As a result of which, both Al2O3 and Fe2O3 react with each other.
⇒ \(2 \mathrm{Al}(s)+\mathrm{Fe}_2 \mathrm{O}_3(s) \rightarrow 2 \mathrm{Fe}(s)+\mathrm{Al}_2 \mathrm{O}_3(s)+\text { Heat }\)
The reduction of Fe2O3 by Al is highly exothermic (2500 – 3000°C), which takes place within a maximum time of 30 seconds.
This produces molten Al2O3 and molten Fe. Molten Fe goes at the bottom since it is heavier than molten Al.
Application: Molten Fe is used for welding ferrous metals like the joining of rails, pipes, and broken parts of large gears.
Activity series of metals: It is a list of metals in ascending order of their activity. From top to bottom, the tendency of gaining e“s decreases i.e. reducing ing property gradually decreases.
Hence, the most reactive metal (K) exists at the top and the least reactive metal (Au) at the bottom. Any metal in this series can displace any other metal below it from its salt solution.
Example: Fe + ZnSO4 does not take place, but \(\mathrm{Zn}+\mathrm{FeSO}_4 \rightarrow \mathrm{ZnSO}_4+\mathrm{Fe}\) Fe can take place as the position of Zn is above Fe in the series.]

Reactive metals like Au and Pt exist in a free state in nature. So, for these metals reduction is not needed.
Oxides/sulphides of Ag, and Hg are less stable. They can be reduced by heat. ng.
Oxides/sulphides of Zn and Fe can only be reduced by using Coke or any other suitable substances.
Oxides of most reactive metals (K, Na, Ca, Mg, Al) are stable and ionic compounds. They can not be reduced using coke or any other reducing substance.
Electrolysis is the only process (applying an electric field or potential difference) -in which reduction takes place at the cathode.
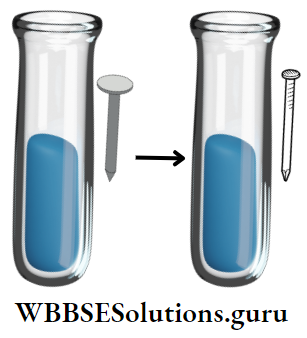
Activity: Carry out the following activities and note down your observations :
Add Cu-wire in ferrous sulphate solution (FeS04) → No reaction.
Add iron nails in copper sulphate solution (CuSO4) → The Blue colour of CuSO4 fades away and brown-coloured FeSO4 deposits on Fe-nails. (Explain the reason)
WBBSE Chapter 8 Metallurgy Metal Corrosion
Usually, metals are lustrous/shining. But with time, metals start appearing dull, less shining. This is because of metal corrosion.
You might have noticed the difference in the appearance of a gold ring, silver spoon, copper coin, iron nail, and aluminium toy left in the air for a long time.
Silver spoon will appear less lustrous having a black coloured layer [Ag + H2S (from the air) → Silver spoon Copper coin Ag2S (black silver sulphide)]
On the copper coin, a green-coloured layer will form, and the iron nail will appear in the worst condition having a reddish-brown layer (rust),
The gold ring will remain in the best condition (as gold is the least reactive), and the aluminium toy will also be preserved very well.
Metals are considered to be corroded when they react with air/water. Corrosion happens very slowly.

Corrosion Definition: Corrosion is described as the formation of compounds on the surface of a metal when it is exposed to air/water or moisture/acid or electrolyte like salt water.
Rusting (Corrosion of iron): If impure iron is left in moist air, it gets corroded with a reddish brown coating on its surface.
This coating is called rust which is a hydrated ferric oxide (Fe2O3 . x H2O where x represents an unknown quantity of water used in rusting and it is a variable) and the entire process is called rusting.
Rust is porous and can be removed. Iron corrodes very easily as compared to other metals like Cu/Ag/AI.
Because Fe is more reactive than Cu/Ag/AI. In this process of rusting, the upper surface of Fe is eaten up gradually.
Rusting weakens Fe objects and cuts short their lives. So, it’s an economic loss also. Pure Fe does not rust.

Mechanism of rusting of iron: For rusting to occur -Fe, water and O2 are required. Here we will study the roles of water and O2 in rusting.
The overall change “Fe→ Fe2+(unstable state) → Fe3+ (very stable state) → Fe2O3 • x H2O¯ is a redox reaction i.e. an irreversible process once the rust is formed, it is not possible to get back the original Fe from rust.
When Fe-surface is exposed to water droplets, the Fe– at- oms at the centre of water droplets give up 2 e¯sto form Fe2+ Fe (s) → Fe2+ (aq.) + 2e– (oxidation takes place – here Fe is oxidised)
The e–s move from the centre of the water droplet towards the edge all around. At the edge of the water droplet, there is an abundance of O2.
O2 is reduced to OH– ions in the presence of water.
⇒ \(\mathrm{O}_2(g)+2 \mathrm{H}_2 \mathrm{O}(\mathrm{I})+4 e^{-} \rightarrow 4 \mathrm{OH}^{-} \text {(aq.) (reduction) }\)
We know that the electrode anode always donates e– s and the cathode gains e– s.
So, on Fe– surface plenty of small local chemical cells are formed, where each Fe atom at the centre of the water droplet acts as an anode,
The edge of the water droplet where C (impurity)-atoms are present acts as the cathode and the water molecule as an electrolyte.
Fe2+ ions react with OH– ions to form solid ferrous hydroxide [Fe (OH)2
⇒ \(\begin{aligned}
& 2\left[\mathrm{Fe} \rightarrow \mathrm{Fe}^{2+}+2 \mathrm{e}^{-}\right] \\
& \frac{\mathrm{O}_2+2 \mathrm{H}_2 \mathrm{O}+4 \mathrm{e}^{-} \rightarrow 4 \mathrm{OH}^{-}}{2 \mathrm{Fe}+2 \mathrm{H}_2 \mathrm{O}+\mathrm{O}_2 \rightarrow 2 \mathrm{Fe}^{2+}+4 \mathrm{OH}^{-}} \rightarrow 2\left[\mathrm{Fe}^{2+}+2 \mathrm{OH}^{-}\right] \rightarrow 2 \mathrm{Fe}(\mathrm{OH})_2 \text { (s) (unstable) } \\
&
\end{aligned}\)
Fe (OH)2 is further oxidised by O2 and forms ferric hydroxide [Fe (OH)3].
⇒ \(4 \mathrm{Fe}(\mathrm{OH})_2(s)+2 \mathrm{H}_2 \mathrm{O}(\mathrm{I})+\mathrm{O}_2(g) \rightarrow 4 \mathrm{Fe}(\mathrm{OH})_3(s) \text { (a stable compound) }\)
Fe (OH)3 decomposes into hydrated ferric oxide.
Fe (OH)3 (s)→Fe2O3 • x H2O (s)
Overall electro-chemical rusting reaction: 4Fe (s) + 2 x H2O (l) + 3O2 (g) 2Fe2O3 • x H2O(s)
Note: Rusting is faster in the presence of chloride (Cl–) ions. It is a serious problem in sea-going ships or submerged parts of pipelines.
Prevention of rusting:
Barrier protection: The simplest way of preventing rusting is by painting/oiling/greasing iron objects because of which iron will not get exposed to moisture.
Metallic coating: Rusting can be prevented by making a coating of another more reactive metal.
For example Galvanization
(Coating of Zn on Fe): Zn is more reactive than Fe. So, Zn reacts more rapidly than Fe forming a thin layer of ZnO on the surface of the Fe-object.
It does not allow more O2 to react with the inner layer of Fe. Thus, galvanization is a better way to prevent Fe from rusting,
Tin plating, Chromium plating (plating of chromium metal Nickel plating This is done by electrolysis.
Alloying: Fe rusts very easily. But when Fe is mixed with Cr and Ni (an alloy), it becomes strong as well as does not rust. (Cr, Ni give a shiny look to Fe).
Cathodic protection: In an electro-chemical reaction, Fe-atoms (anode) at the surface give up e“s which flow towards the cathode because of this the anode gets corroded slowly.
In the cathodic protection method, the flow of e”s can be stopped by the use of (a) a sacrificial anode.
A more active metal like Mg-block is connected with Fe so that Mg acts as an anode instead of Fe. So, oxidation takes place in Mg.
[Mg (s)→ Mg2+ (aq.) + 2e ]. Instead of Fe- Mg gets corroded. This process is used for the protection of underground pipes, tanks etc.
When the Mg-block is corroded completely, it has to be replaced by a fresh one.
Sherardizing is the process of formation-corrosion-resistant Zn-layer by vapour galvanizing on the surface of iron or steel.
The Delhi Pillar of the Gupta Age is a unique metallurgical marvel of high-quality of steel production in ancient India. It was made from 98% corrosion-free wrought iron.
Corrosion of other metals and its health implications:
Al is a good conductor of heat and electricity. It is not affected in dry air. But in contact with moist air, Al reacts with the O2 from air forming aluminium oxide (Al2O3) which is very much unreactive.
Once the Al2O3 layer is formed, it binds very tightly with the Al-surface. As a result of which, the thermal and electrical conductivity of Al decreases. Although, the Al2O3 layer keeps Al unaffected in moist air.
We have learnt that metals get corroded in moist air for a long time. On Cu or alloys of Cu, left in the open air for many years, green-coloured patches are formed. CO2 and water vapour reacting with Cu form basic copper carbonate [Cu + O2 + CO2 + H2O →Cu (OH)2 • CuCO3].
Al, Zn react with weak organic acidic substances like vinegar or lemon juice, and they react to form soluble metallic compounds (harmful to our health).
Cu also forms soluble metallic compounds by reacting with acidic pickles or fruits. For this reason, acidic foods should not be kept or processed in Al, Zn or Cu containers.
Also, tarnished metallic utensils should be cleaned well before use to avoid metal poisoning.

