WBBSE Chapter 8 Electricity And Chemical Reactions Electrolytes
Electrolytes Definition: Electrolytes are the chemical compounds that ionize or dissociate into their ions [cations (+) and anions (-)] in a molten/aqueous state and the solutions so-produced have the ability to conduct electricity (d.c.).
Take some table salt (NaCI, an ionic compound) and heat over 801°C, it melts and the ions Na+ and CP already present in it, get released.
Similarly, in its aqueous solution, H2O molecules occupy some positions of NaCI molecules and because of the dielectric property of water, the electrostatic force of attraction between Na+ and Cl” ions is decreased so the ions get free.
Ionic compounds (NaCI, KCI, CuSO4, CuCI2, NaOH, etc.) in a molten/aqueous state can conduct electricity through these ions.
They behave like conductors but are different from metals. But solid NaCI is not an electrolyte. Because in the solid state, a strong electrostatic force of attraction exists between the Na+ and CP ions the ions are not free to move.
WBBSE Notes For Class 10 Physical Science And Environment
In the case of covalent compounds like HCI (g), no ions exist but in an aqueous solution, it forms HCI (strong acid) which can be dissociated into H+ and CP ions. This is called ionization. Other such examples are HF, HBr, HI, NH3, etc.
Also, there are some covalent compounds like (pure) distilled water, sugar solution, alcohol, ether, benzene, kerosene, molten sulfur, ethanol, urea, and glycerine which do not dissociate into ions and cannot conduct electricity even by little amount.
They are called nonelectrolytes. They behave like non-conductors of electricity because they do not belong to free electrons (like metals) and ions (like electrolytes).

WBBSE Chapter 8 Electricity And Chemical Reactions Strong And Weak Electrolytes
This classification is based on the degree of ionization or the efficiency of dissociation that an electrolyte exhibits in its aqueous solution.
A greater degree of ionization corresponds with a stronger electrolyte. We can classify strong electrolytes, weak electrolytes, and non-electrolyte by measuring the electrical conductivity of their aq. solutions.
See the experimental-
Observation of the experiment: Aq. solution of NaCI or KCI or H2SO4 or NaOH can conduct electricity very well so they are strong electrolytes.
On the other hand, acetic acid CH3COOH aq.) or ammonium hydroxide (aq.) can also conduct electricity but not as well as a strong electrolyte they are weak electrolytes.
The non-electrolytes like ethanol/ benzene/ glycerine show zero conductivity.
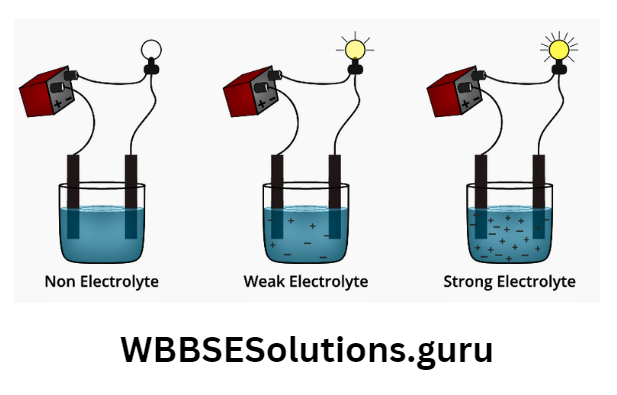
Strong electrolyte: If a substance is molten/aq. the state completely dissociates into ions such that the solution consists entirely of ions, and molecules left, and can conduct electricity very well is called a strong electrolyte.
Strong acids (H2S04, HN03, HCI), strong soluble bases (NaOH, KOH), and soluble salts (NaCI, K2S04, CuS04, CuCI2) are strong electrolytes.
Ionization reaction:
⇒ \(\mathrm{NaCl} \text { (aq.) } \rightarrow \mathrm{Na}^{+}+\mathrm{Cl}^{-} \text {. } \mathrm{CuSO}_4 \text { (aq.) } \rightarrow \mathrm{Cu}^{2+}+\mathrm{SO}_4^{2-} . \mathrm{H}_2 \mathrm{SO}_4 \text { (aq.) } \rightarrow 2 \mathrm{H}^{+}+\mathrm{SO}_4{ }^{2-} \text {. }\)
Since every molecule of a strong electrolyte dissociates into ions, so a single arrow (→) is used in the dissociation reaction. It’s a chemical change.
Weak electrolyte: If a substance in aq. solution partially ionizes (which means only a few molecules break into ions and most stay as neutral molecules) which still can conduct electricity but not as well as a strong electrolyte then it is called a weak electrolyte.
Weak acids (CH3COOH, H2C03), weak bases (NH4OH), and some salt (AgCI) are examples.
Ionization reaction:
⇒ \(\mathrm{CH}_3 \mathrm{COOH} \text { (aq.) } \rightleftharpoons \mathrm{H}^{+}+\mathrm{CH}_3 \mathrm{COO}^{-} . \mathrm{NH}_4 \mathrm{OH} \text { (aq.) } \rightleftharpoons \mathrm{NH}_4^{+}+\mathrm{OH}^{-} .\)
In such solutions, an equilibrium is established between the ions and the non-ionized neutral molecules.
This is represented by a double arrow (![]() ) between the ions and the non-ionized molecules of weak electrolytes.
) between the ions and the non-ionized molecules of weak electrolytes.
WBBSE Chapter 8 Electricity And Chemical Reactions Mechanism Of Electrical Conduction In Molten Solution State
Take molten/aq. solution of table salt (NaCI) in a glass bowl. In the solution state, NaCI contains plenty of Na+ (cation) and Cl– (anion) ions. \(\left[\mathrm{NaCl} \text { (aq.) } \rightarrow \mathrm{Na}^{+}+\mathrm{Cl}^{-}\right]\)
Two conducting rods (electrodes) are inserted into the solution.
The rod connected with the + ve terminal of the battery is the anode (A) and the rod connected with the – ve terminal of the battery is the cathode (C).
[The electrodes are usually made of inert material (platinum/graphite) to ensure that the electrodes itself do not involve in the electrolytic reactions.] Now let us see what happens when the electrodes (say 9V or 12V).
⇒ \(2 \mathrm{NaCl} \text { (aq.) } \stackrel{\text { electricity }}{\longrightarrow} 2 \mathrm{Na}^{+}+2 \mathrm{Cl}^{-} \text {. }\)

The Na+ ions get attracted towards the – ve electrode (cathode) and Cu ions are attracted by the +ve electrode (anode).
Thus, the cations and anions move in opposite directions for the purpose of electrical discharge.
At the cathode, each Na+ ion captures le_ to produce metal Na (it is a reduction reaction) and at the anode, each Cl¯ ion gives up le¯ and becomes neutral Cl.
Atom (it is an oxidation reaction). The single Cl atoms then pair up and form Cl2 gas. Electron flows from the anode to the cathode in the external circuit.
At the Cathode:
⇒ \(\begin{array}{r}
2 \mathrm{Na}^{+}+2 e^{-} \rightarrow 2 \mathrm{Na}(s) \ldots . . \\
\quad \text { (reduction reaction) }
\end{array}\)
At the anode:
⇒ \(2 \mathrm{Cl}^{-} \rightarrow 2 \mathrm{Cl}+2 e^{-}, 2 \mathrm{Cl} \rightarrow \mathrm{Cl}_2(g)….. (oxidation reaction)\)
So, solid sodium is formed at the cathode, and chlorine gas being given released at the anode (chemical change).
Here, the coefficients used in the equation (for overall electrical neutrality of the entire system) indicate mol numbers.
In this reaction, at the anode 2 mol Cl– ion removes 2 mol e¯, and finally, 1 mol Cl2 gas is produced. Similarly, at the cathode 2 mol Na+ ion gain 2 mol e¯, and finally, 2 mol Na (s) is produced.
That’s why, the mechanism of electrical conduction metals and electrolytes are not exactly the same.
In electrical conduction through molten/aq. electrolytes only the anions and cations (not electrons like conductors) take part, and during electrolysis, they move in opposite directions.
The difference in electrical conductivity between metals and electrolytes:

WBBSE Chapter 8 Electricity And Chemical Reactions Electrolysis
Electrolysis Definition: Electrolysis is a chemical dissociation/ionization of an electrolyte (chemical change) in a molten/aqueous state by passing direct current (d.c.)-
Electrolysis Example:
⇒ \(\mathrm{CuSO}_4 \text { (aq.) } \stackrel{\text { electricity }}{\longrightarrow} \mathrm{Cu}^{2+}+\mathrm{SO}_4^{2-} ; \quad \mathrm{HCl} \text { (aq.) } \stackrel{\text { eléctricity }}{\longrightarrow} \mathrm{H}^{+}+\mathrm{Cl}^{-}\)
The ionic compounds in their solution state, ionize into cations and anions which conduct electricity.
In the case of covalent compounds like HCI (g)- no free ions are there, which in an aqueous state dissociates into H+ and Cl¯ ions.
In the case of electrolysis, electrical energy → is chemical energy. Just the opposite for an electric cell where chemical energy → electrical energy.
In the case of electrolysis, the source of electricity must be d.c. (obtained from a cell/battery of low voltage). No heavy current is to be used. No. a.c.
Electrolysis is a type of Redox reaction. Always oxidation (loss in e¯ anion) takes part at the anode and reduction (gain in e¯ by cation) at the cathode.
With respect to electron transfer, the cathode can be defined as the electrode towards which cations move and the anode is the electrode towards which anions move.
Electrolysis of acidified water using Pt-electrode: Already learned that pure water has no free ions to conduct electricity. Few drops of oil. H2SO4 can ionize pure water.
Then pure water becomes to be an electrolyte.
Electrolytic cell: Electrolysis of acidified water forms a cell, called Hoffman Voltameter.
The arrangement in which two Pt-electrodes and a 12 V d.c. supply batteries are used. (Pt is inert and cannot react with the solution).
Ionization:
⇒ \(\mathrm{H}_2 \mathrm{O}(\mathrm{H}-\mathrm{OH}) \rightarrow \mathrm{H}^{+}+\mathrm{OH}^{-}, \mathrm{H}_2 \mathrm{SO}_4 \rightarrow 2 \mathrm{H}^{+}+\mathrm{SO}_4{ }^{2-}\)
When more than one anions/cations come into the electrolytic solution, then only one of them gets preferentially discharged at the electrode.
This factor depends on the position of anion/cation in the electrochemical series, from which we can determine the tendency to lose/ gain e¯s.
This tendency is as follows:
At Cathode:
⇒ \(\begin{aligned}
& \mathrm{Ag}^{+}>\mathrm{Cu}^{2+}>\mathrm{H}^{+}>\mathrm{Fe}^{2+}>\mathrm{Zn}^{2+}>\mathrm{Al}^{3+}>\mathrm{Mg}^{2+} \\
& >\mathrm{Na}^{+}>\mathrm{Ca}^{2+}
\end{aligned}\)
At anode:
⇒\(\mathrm{I}^{-}>\mathrm{Br}^{-}>\mathrm{Cl}^{-}>\mathrm{OH}^{-}>\mathrm{NO}_3^{-}>\mathrm{SO}_4^{2-}>\mathrm{F} .\)
As soon as the switch is on, a lot of bubbles are seen to be formed in two tubes.
This happens because – both the an-ions OH and SO42¯ get attracted towards the anode while cations H+ are attracted by the cathode.
According to the electrochemical series, OH¯ has a greater tendency to lose e” than SO42¯. Thus, at the anode, each OH¯ ion loses le¯ (oxidation), and at the cathode, each H+ ion gains le¯ (reduction).
So electron flow continues from anode to cathode in the external circuit and constitutes a flow of electric current from anode to cathode in the electrolyte solution.
At anode:
⇒ \(\begin{aligned}
& \mathrm{OH}^{-} \rightarrow 1 e^{-}+\mathrm{OH} \text { (unstable); } \mathrm{OH}+\mathrm{OH} \rightarrow \mathrm{H}_2 \mathrm{O}+\mathrm{O} \text { (nascent); } \mathrm{O}+\mathrm{O} \rightarrow \mathrm{O}_2 \\
& 4 \mathrm{OH} \rightarrow 4 e^{-}+4 \mathrm{OH} ; 4 \mathrm{OH} \rightarrow 2 \mathrm{H}_2 \mathrm{O}+\mathrm{O}_2 \uparrow \text { (oxidation) }
\end{aligned}\)
At Cathode:
⇒ \(\begin{aligned}
& \mathrm{H}^{+}+1 e^{-} \rightarrow \mathrm{H} ; \mathrm{H}+\mathrm{H} \rightarrow \mathrm{H}_2 \\
& 4 \mathrm{H}^{+}+4 e^{-} \rightarrow 2 \mathrm{H}_2 \uparrow \text { (reduction) }
\end{aligned}\)
Observation:
At the – ve and + ve electrodes, the gases collected are detected as H2 and O2 respectively. The ratio of the volume of H2 and O2 is 2 :1.
Electrolysis of CuSO4 (aq.) using inert (Pt/graphite) electrode: Two Pt-electrodes are used as anode (+ ve) and cathode (- ve).
In place of Pt, graphite can also be used. Pt. is nonreactive but there is a little chance of oxidation of graphite.
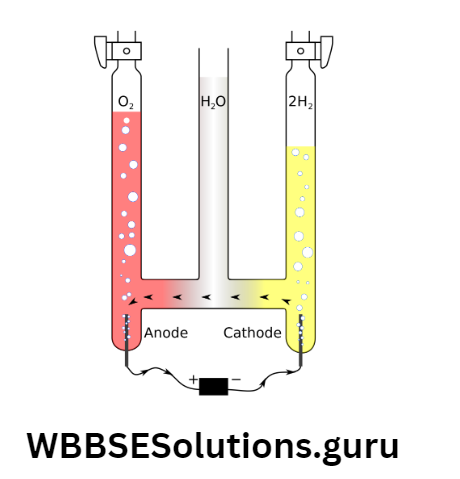
Ionization: \(\mathrm{CuSO}_4 \text { (blue solution) } \rightarrow \mathrm{Cu}^{2+}+\mathrm{SO}_4^{2-}, \quad \mathrm{H}_2 \mathrm{O}(\mathrm{H}-\mathrm{OH}) \rightarrow \mathrm{H}^{+}+\mathrm{OH}^{-}\)
As soon as the switch is on, the -vely charged OH¯ ions move towards the anode (+ ve), and + very charged Cu2+ ions move towards the cathode (- ve).
Because according to the electrochemical series, OH¯ has a greater tendency to lose e¯ then SO42+, and Cu2+ has a greater tendency to gain e¯ than H.
Then OH¯ and Cu2+ ions are discharged at the anode and cathode respectively.
At Anode: \(\mathrm{OH}^{-} \rightarrow 1 e^{-}+\mathrm{OH} ; 4 \mathrm{OH}^{-} \rightarrow 4 e^{-}+4 \mathrm{OH} ; 4 \mathrm{OH} \rightarrow 2 \mathrm{H}_2 \mathrm{O}+\mathrm{O}, \uparrow \text { (oxidation) }\)
At Cathode: \(\mathrm{Cu}^{2+}+2 e^{-} \rightarrow \mathrm{Cu} \downarrow ; 2 \mathrm{Cu}^{2+}+4 e^{-} \rightarrow 2 \mathrm{Cu} \downarrow \text { (reduction) }\)
Observations :
Products are O2 gas at the anode and Cu deposited at the cathode.
Pink/reddish brown Cu metal gets deposited at the cathode. So, cathode mass increases.
The presence of Cu2+ ions makes CuSO4 While electrolysis, these Cu2+ ions from the solution get deposited slowly at the cathode, so the blue color of CuSO4 solution fades away.
Electrolysis of CuSO4 (aq.) using Cu electrodes:
In the Previous case, we used Pt-electric rods in the case, and both the electrodes are of Cu.
In this case, both electrodes are of Cu. Now let’s see what difference is observed in the electrolysis of CuSO4 solution.
Now let’s see what difference is observed in the electrolysis of CuSO4 solution.
The ions present in the solution are
⇒\(\mathrm{Cu}^{2+}+\mathrm{SO}_4^{2-}\mathrm{H}^{+}, \mathrm{OH}^{-}\)
At cathode: \(\mathrm{Cu}^{2+} \text { (aq.) }+2 e^{-} \rightarrow \mathrm{Cu}(s) \text { [Reduction reaction] }\)
So Cu2+ ions on being discharged are deposited at the cathode as Cu-atoms.
At anode: \(\mathrm{Cu}(\mathrm{s}) \rightarrow \mathrm{Cu}^{2+}+2 e^{-} \text {[Oxidation reaction] }\)
Within the solution, Cu2+ ions exist and the anode itself is made up of Cu-atoms. In such a case, neither OH+ nor SO42+ will be discharged – rather, at the anode each Cu-atom losing 2e¯s goes into the solution as a Cu2+ ion.
As a whole, when a Cu2 ion is deposited at the cathode, simultaneously from the anode another Cu2+ ion goes into the solution.
Ultimately, the amount by which anode mass decreases, and cathode mass increases by the same amount.
Also, the concentration of CuSO4 solution effectively remains constant. So, the blue color of the CuSO4 solution doesn’t fade away. (This process is used as electroplating).
WBBSE Chapter 8 Electricity And Chemical Reactions Applications Of Electrolysis
Electrolysis is mostly used in industry in many ways such as
In metal extraction,
In electrorefining,
In electroplating.
Electrolysis in the extraction of metals from their ores: Highly electropositive (active) metals like Na, Ca, and Al are extracted from their ores using electrolysis.
Al-Extraction: The principal ore of Al is bauxite (Al2O3-2H2O).
It is purified to yield aluminum oxide (Al2O3) [white powder – its m.p. is very high (over 2000°C)].
Al2O3 is dissolved in molten cryolite (Na3AlF6) [an Al-compound with much lower m.p. than Al2O3 and the temperature is maintained at ~ 925°C.
Molten Al2O3 contains Al-ions (Al3+) and Oxide-ions (O2- ).
Both the electrodes are made up of graphite. More than one graphite rod is used as the anode.
At the Anode: \(\mathrm{O}^{2-}-2 e^{-} \underset{\text { oxidation }}{\longrightarrow} 0 ; 2 \mathrm{O}^{2-} \rightarrow \mathrm{O}_2(g)+4 e^{-}\)
At the Cathode: \(\mathrm{Al}^{3+}+3 e^{-} \underset{\text { Reduction }}{\longrightarrow} \mathrm{Al}(\mathrm{s})\)
The O2 molecules formed at the anode react with the graphite (carbon atoms) rods (Anode) and form \(\mathrm{CO}_2 \text { gas. }\left[\mathrm{C}+\mathrm{O}_2 \rightarrow \mathrm{CO}_2(g)\right] \text {. }\)Due to this reason, the anode rods are regularly replaced.
Electro-refining of copper: Electro-refining is a process of purifying an impure metal to a high purity level to get pure metal using electrolysis.
Electrolytic solution: Aq. CuSO4 + dil. H2SO4.
Cathode: A thin sheet of pure Cu.
Anode: Impure sheet of Cu. The ions present in the solution are Cu2+, SO42-.
At the anode: From impure sheet \(\mathrm{Cu}(\mathrm{s}) \underset{\text { oxidation }}{\longrightarrow} \mathrm{Cu}^{2+}(\mathrm{aq} .)+2 e^{-}\)
At the cathode:
⇒ \(\mathrm{Cu}^{2+}(\text { aq. })+2 e^{-} \underset{\text { Reduction }}{\longrightarrow} \mathrm{Cu}(\mathrm{s})\)
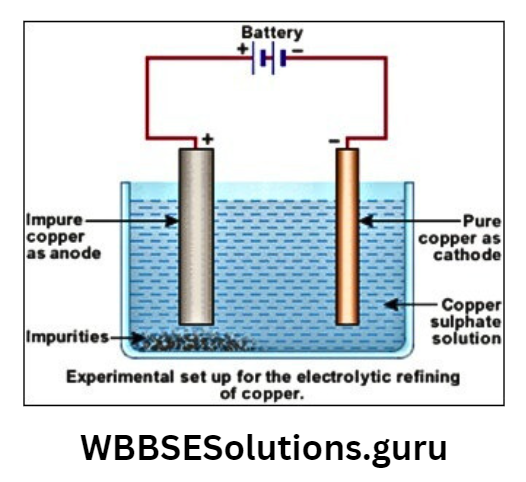
The Cu2+ ions move between the anode and the cathode. When a Cu2+ ion is deposited cathode, at the same time, another Cu ion from an impure Cu sheet goes into the solution.
Thus, the concentration of CuSO4 solution effectively remains the same.
As this happens, the size of the cathode sheet gets larger while the size of the anode sheet gradually decays, a sludge is left at the bottom of the anode which is called anode mud.
The anode als mud (Ag/Au) may contain many valuable met- Cu obtained from electro-refining and is about 99-99% pure.
Electroplating: Electroplating is an electrolytic process by which a thin layer of superior metal or noble metal like gold, silver, nickel, or chromium is deposited/coated on the surface of a base metal either for protection against corrosion or making attractive.
Basic principles of electroplating:
The article to be electroplated is cleaned first,
Metal on which electroplating is to be done is used as the cathode (- ve electrode). Because deposition always takes place at the cathode,
A pure block of the metal to be electroplated is used as the anode (+ ve electrode),
A solution of a salt of the metal is used as the electrolyte (the solution contains metal ions).
For example, in Cu-plating the electrolyte is CuS04(aq.); in silver plating, silver nitrate (AgNO3)/potassium argento cyanide K [Ag (CN)2J solution;
In gold-plating, potassium auro cyanide K [Au (CN)2] solution; in nickel plating, a mixed solution of
⇒ \(\mathrm{NiSO}_4+\left(\mathrm{NH}_4\right)_2 \mathrm{SO}_4+\text { slight boric acio }\)
For getting uniform deposition, a low direct current (3A or 4A) for a longer time is passed through the solution. (No heavy current to be used or no a.c. to be used).
